Quality of life measures in sino nasal disease
Chronic rhinosinusitis (CRS) is a chronic inflammatory disease that affects the nasal and sinus cavities. To measure the quality of life (QoL) in patients with CRS, researchers typically use disease-specific QoL instruments that are designed to capture the impact of the disease on the patient’s physical, emotional, and social well-being.
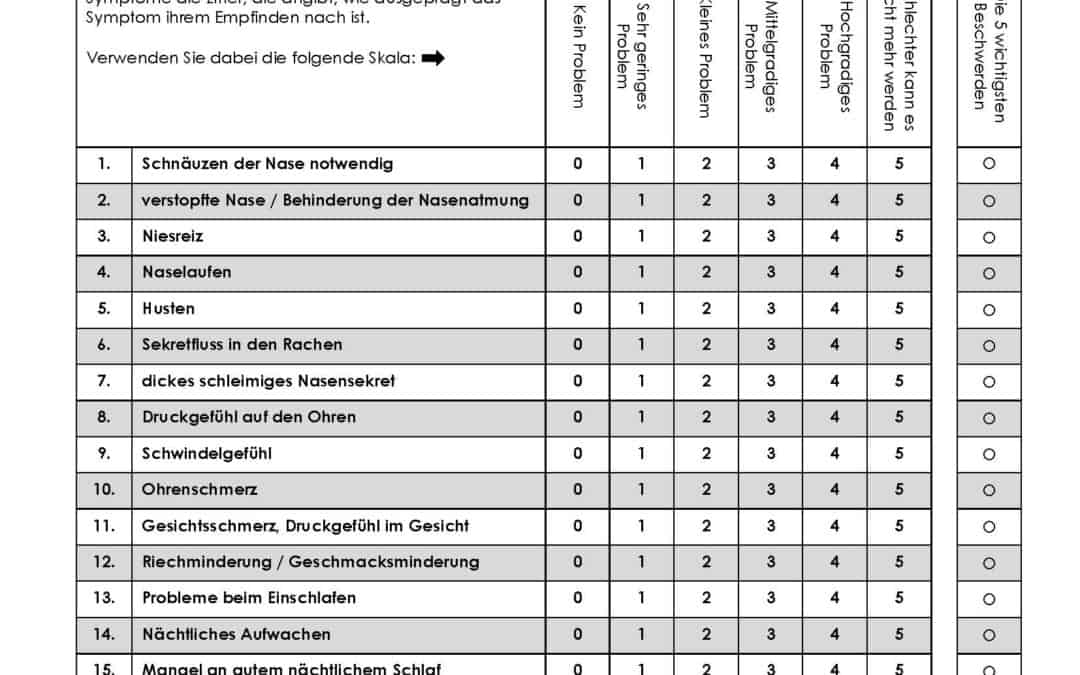
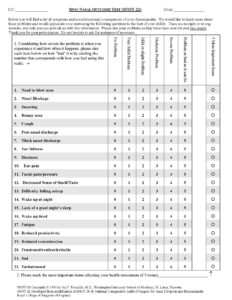
The most commonly used instrument for measuring QoL in patients with CRS is the Sino-Nasal Outcome Test (SNOT), which was developed by the Rhinology Research Society. The SNOT 22 questionaire is a validated instrument that asks patients to rate their symptoms, such as nasal obstruction, facial pain, and postnasal drip, on a scale from 0 (no problem) to 5 (problem as bad as it can be). The questionnaire also includes items that assess the impact of CRS on the patient’s daily activities and emotional well-being.
Other disease-specific QoL instruments that are used to measure the impact of CRS on QoL include the Rhinosinusitis Disability Index (RSDI), the Sinonasal Outcome Test-22 (SNOT-22), and the Chronic Sinusitis Survey (CSS). These instruments assess various aspects of QoL, including symptoms, functional limitations, emotional well-being, and social functioning.
In addition to disease-specific QoL instruments, researchers may also use generic QoL instruments to measure the impact of CRS on overall QoL. The most commonly used generic QoL instrument is the Short Form-36 (SF-36), which assesses overall QoL and includes domains such as physical functioning, social functioning, and emotional well-being.
Overall, the choice of QoL instrument used to measure the impact of CRS on QoL may depend on the specific research question, the patient population, and the clinical setting. However, the most commonly used instruments are disease-specific QoL instruments, such as the SNOT 22 questionnaire, which are designed to capture the unique impact of CRS on QoL. Currently, despite some limitations, the SNOT22 are the most widely accepted and researched quality of life tool in chronic rhinosinusitis. The questionaire has also been validated in other sino-nasal disorders. An example of the English version is featured below.
SNOT 22 questionaire translations are availiable for clinical practice and research
To date, the SNOT 22 questionaire has been translated into more than 40 languages, including French, German, Spanish, Chinese, and Korean. These translations have been conducted using established translation and cross-cultural adaptation processes to ensure the validity and reliability of the translated instrument.
Several of these translations have been validated in clinical studies, demonstrating their utility in assessing the impact of CRS on quality of life in different populations. The availability of these translated versions of the SNOT 22 questionaire has facilitated the use of this instrument in international clinical research, allowing for cross-cultural comparisons of CRS outcomes.
In summary, there are more than 40 translations of the Sino-Nasal Outcome Test, which have been validated and used to assess the impact of chronic rhinosinusitis on quality of life in diverse populations around the world.
Our links to the SNOT22 questionaire PDFs for download (for research use):
- English
- German
- French
- Arabic
- Brazil – Portugese
- Philipines – Tagalog
- Greek
- Hebrew
- Thai
- Turkish
- Chinese
SNOT22 test characteristics:
Scale 0-5 or 0-110
MCID: 9.8
Subdomains: Nasal, Ear/Facial, Sleep, Function, Emotion
General asymptomatic population: 20 and below