Remission in Chronic Rhinosinusitis with Nasal Polyps (CRSwNP): Why “Feeling Better” Is No Longer Enough
For years, success in chronic rhinosinusitis with nasal polyps (CRSwNP) meant one thing: the patient feels better.
But in 2025, that is no longer our goal.
With the arrival of biologic therapies for nasal polyps—including dupilumab, mepolizumab, tezepilumab and benralizumab—the target has shifted. In other airway diseases, especially asthma, the conversation has moved beyond symptom improvement toward a more ambitious endpoint: remission. Severe asthma remission has been defined as a composite state (no exacerbations, no systemic corticosteroids, sustained symptom control, stable objective measures, over time).(1) Real-world studies show that remission is achievable in a meaningful subset of biologic-treated asthma patients.(2,3)
CRSwNP shares overlapping type 2 inflammatory biology with asthma and responds to targeted biologics.(4–7) So the obvious question is:
Should we aim for remission in nasal polyps too?
The answer is yes—but CRS is structurally different.
Why Sinus Surgery Changes the Remission Conversation
Unlike asthma, we can surgically modify the anatomy in CRSwNP.
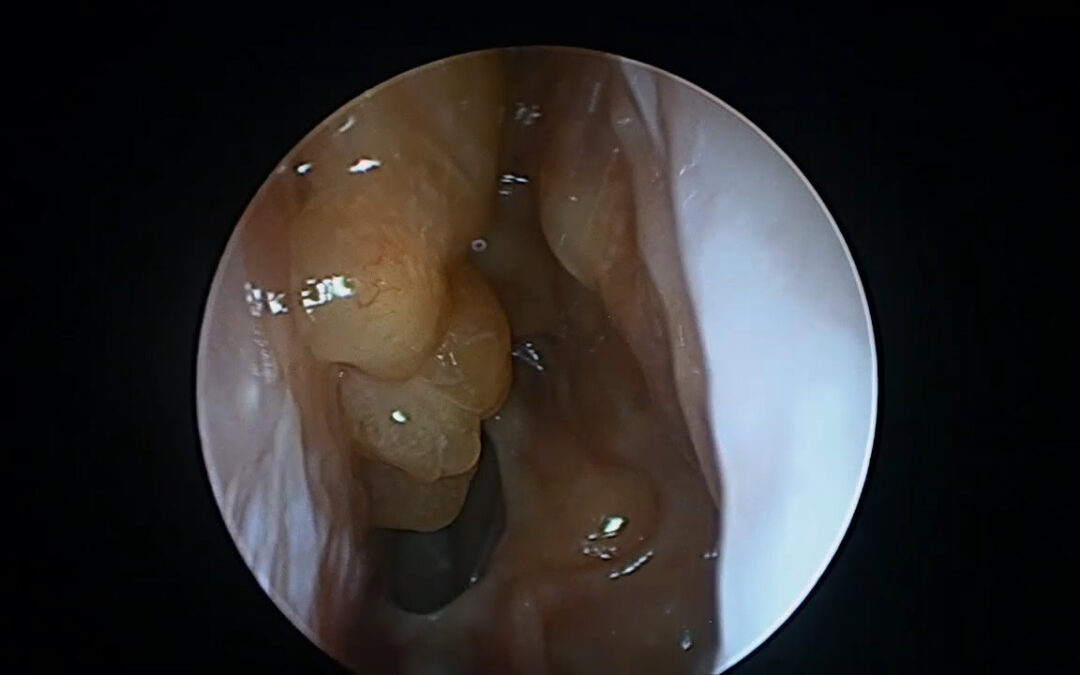
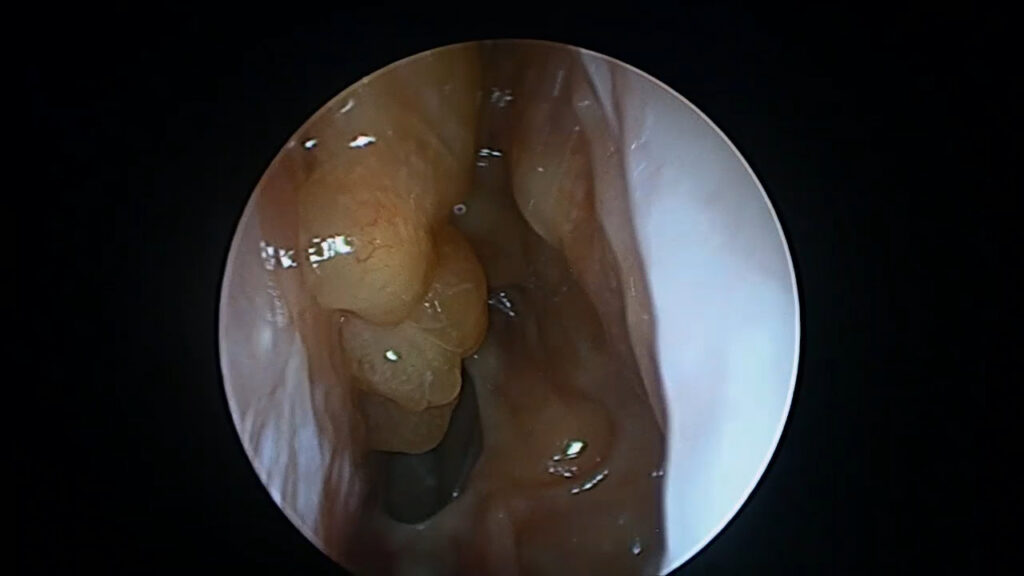
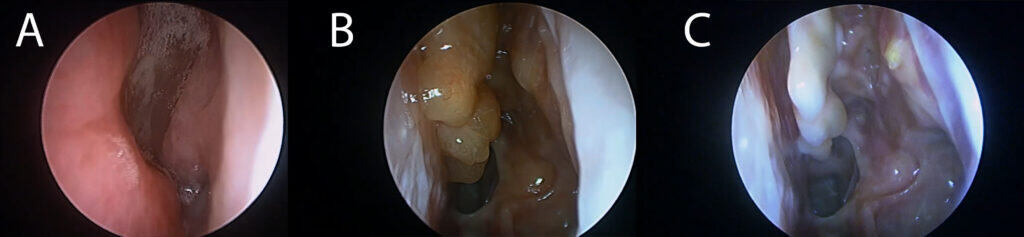
Procedures such as complete sphenoethmoidectomy and extended surgery (e.g., Draf IIc/III) reshape the inflammatory environment, improve topical access, and reduce persistent diseased compartments.(8–10) Distribution studies demonstrate that extended frontal procedures can improve irrigation penetration compared with more limited frontal approaches.(9)
Recent evidence also shows that surgical completeness predicts disease trajectory and may influence response to subsequent biologic therapy. CT-based measures of completeness such as ACCESS (Amsterdam Classification of Completeness of Endoscopic Sinus Surgery) quantify how complete surgery is and have been linked to outcomes.(11) Similarly, the Completion of Surgery Index (CoSI) provides another structured approach to capturing adequacy of surgical extent.(12)
Our recent study—Extent of sinus surgery is associated with disease control in biologic treated type-2 dominant CRS—found that extended neo-sinuis surgery was strongly associated with achieving disease control in biologic-treated patients.(13) Clincial remisison achieved in 60%+ of patients.
In other words: surgical completeness matters.
So remission in CRSwNP cannot be defined by symptoms alone.
CRSwNP Remission vs. Symptom Improvement
Traditionally, studies report:
-
Change in SNOT-22
-
Change in polyp score
-
Corticosteroid use
-
Avoidance of revision surgery
These are essential metrics and remain central in consensus guidance.(14,15) But they measure improvement, not a stable disease state.
A patient can be polyp-free yet still require repeated systemic corticosteroids. That is not remission.
That is partial control.
What Should Remission in Nasal Polyps Look Like?
A practical remission framework for CRSwNP should include multiple domains, similar to other inflammatory diseases:
Complete Clinical Remission (≥12 months)
-
SNOT-22 <20 (supported by SNOT-22 validation work)(16)
-
No systemic corticosteroids
-
No polyp recurrence
-
Stable endoscopic findings
-
Stable surgical cavity
Functional Remission
-
SNOT-22 <30 (pragmatic threshold used in clinical response frameworks)(17)
-
Systremic corticosteroid independence
-
Minimal, non-progressive inflammation (on endoscopy)
This tiered model mirrors the concept of strict vs pragmatic remission used in other biologic-era disease frameworks.(1)
SNOT-22 <20 or <30? Why This Debate Matters
The SNOT-22 score is central for capturing symptom burden and quality of life.(16)
-
<20 approximates near-normal burden.
-
<30 reflects low residual burden and is used in response frameworks for biologic monitoring.(17)
But many severe CRSwNP patients have persistent smell loss, sleep disturbance, comorbid asthma, and long-standing remodeling—even when inflammation is controlled.(14,15)
A pragmatic solution is:
-
<20 for “complete remission”
-
<30 for “functional remission”
Steroid-Free Remission: The Non-Negotiable Goal
Systemic corticosteroids remain a marker of uncontrolled inflammation and cumulative harm.
True remission in CRSwNP should require:
-
No oral corticosteroid bursts
-
No long-term steroid dependence
This is consistent with remission principles in asthma frameworks.(1–3)
The Economics of Biologics vs Surgery
Biologic therapy for nasal polyps is highly effective—but expensive.
Cost-utility analyses have shown major long-term cost differences between biologic strategies and surgery.(18) Budget impact analyses suggest biologic-first strategies can dramatically increase direct costs in uncontrolled CRSwNP.(19)
If optimized surgery improves the likelihood of durable control, then sequencing and integration matter.
What This Means for Patients With Nasal Polyps
Patients often ask: “Is this cured?”
CRSwNP is chronic, and cure remains unlikely for many.
But remission is increasingly achievable.
And remission means:
-
No oral corticosteroids
-
No flares
-
Stable sinuses
-
Durable quality of life
-
No progressive inflammation
That is different from temporary symptom relief.
The Future: Real-World Remission Data
Asthma has registry-scale remission reporting.(3)
CRSwNP needs the same.
That is changing and our recent data suggests that a combination of complete surgery, careful biologic selection and phenotyping will acheive remission in 60%+ (13)
The next decade of rhinology should not focus only on polyp shrinkage, removal or management.
It should focus on:
-
Corticosteroid-free remission
-
Surgical-stratified outcomes
-
Endotype-guided biologic selection
-
Durable disease control
Frequently Asked Questions About CRSwNP Remission
1. What does remission mean in chronic rhinosinusitis with nasal polyps (CRSwNP)?
Remission in CRSwNP means sustained control of inflammation. It includes no systemic steroid use, no polyp recurrence, stable sinus anatomy, and low symptom burden maintained over time — not just temporary improvement.
2. Is remission the same as cure for nasal polyps?
No. CRSwNP is a chronic inflammatory disease. Remission means the disease is well controlled and inactive, but ongoing monitoring or treatment may still be required.
3. Can biologics put nasal polyps into remission?
Yes. Biologic therapies such as dupilumab, mepolizumab, tezepilumab and benralizumab can significantly suppress type 2 inflammation and may allow patients to achieve steroid-free disease control. Remission is more likely when surgery has optimized sinus anatomy.
4. Does sinus surgery improve remission rates in CRSwNP?
Evidence suggests that more complete sinus surgery — particularly extended procedures that produce a neo-sinus cavity — improves disease control and may enhance response to biologic therapy.
5. What is steroid-free remission?
Steroid-free remission means a patient no longer requires oral corticosteroids to control inflammation and maintains stable symptoms and endoscopic findings over time.
6. What SNOT-22 score defines remission?
A SNOT-22 score below 20 may define “normal”, while a score below 30 may define functional remission with minimal symptom burden.
7. Should surgery or biologics come first in CRSwNP treatment?
Treatment sequencing depends on disease severity and anatomy. Optimized surgery may improve long-term disease control and biologic effectiveness, particularly in type 2 dominant CRSwNP.
References
-
Menzies-Gow A, Bafadhel M, Busse WW, et al. An expert consensus framework for asthma remission as a treatment goal. J Allergy Clin Immunol. 2020;145(3):757-765.
-
Sposato B, Bianchi F, Ricci A, et al. Clinical asthma remission obtained with biologics in real life: patients’ prevalence and characteristics. J Pers Med. 2023;13:1020.
-
Scelo G, et al. On-treatment clinical remission of severe asthma with real-world longer-term biologic use. Eur Respir J. 2023.
-
Bachert C, Han JK, Desrosiers M, Hellings PW, Amin N, Lee SE, et al. Efficacy and safety of dupilumab in patients with severe chronic rhinosinusitis with nasal polyps (LIBERTY NP SINUS-24 and LIBERTY NP SINUS-52). Lancet. 2019;394(10209):1638-1650.
-
Han JK, Bachert C, Fokkens W, Desrosiers M, Wagenmann M, Lee SE, et al. Mepolizumab for chronic rhinosinusitis with nasal polyps (SYNAPSE): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Respir Med. 2021;9(10):1141-1153.
-
Bachert C, Han JK, Desrosiers MY, Gevaert P, Heffler E, Hopkins C, et al. Efficacy and safety of benralizumab in chronic rhinosinusitis with nasal polyps: a randomized, placebo-controlled trial. J Allergy Clin Immunol. 2022;149(4):1309-1317.e12.
-
Meier EC, Schmid-Grendelmeier P, Steiner UC, Soyka MB. Real-life experience of monoclonal antibody treatments in chronic rhinosinusitis with nasal polyposis. Int Arch Allergy Immunol. 2021;182(8):736-743.
-
Bassiouni A, Wormald PJ. Role of frontal sinus surgery in nasal polyp recurrence. Laryngoscope. 2013;123(1):36-41.
-
Barham HP, Hall CA, Hernandez SC, Zylicz HE, Stevenson MM, Zito BA, et al. Impact of Draf III, Draf IIb, and Draf IIa frontal sinus surgery on nasal irrigation distribution. Int Forum Allergy Rhinol. 2020;10(1):49-52.
-
Blauwblomme M, Georgalas C, Ahmed S, Alobid I, Battaglia P, Castelnuovo P, et al. Expert consensus on surgical management of primary diffuse type 2-dominant chronic rhinosinusitis. Int Forum Allergy Rhinol. 2025;15(3):303-316.
-
Reitsma S, Adriaensen G, Cornet ME, van Haastert RM, Raftopulos MH, Fokkens WJ. The Amsterdam Classification of Completeness of Endoscopic Sinus Surgery (ACCESS): a new CT-based scoring system grading the extent of surgery. Rhinology. 2020;58(6):538-543.
-
Workman AD, Kuppusamy K, Lerner DK, Bosso JV, Douglas JE, Kohanski MA, et al. Assessing adequacy of surgical extent in CRSwNP: The Completion of Surgery Index. Int Forum Allergy Rhinol. 2025;15(1):9-17.
-
Campion NJ, Sacks PL, Png LH, Hua E, Darbari Kaul R, Alvarado R, Choy C, Sacks R, Earls P, Campbell RG, Kalish L, Harvey RJ. Extent of sinus surgery is associated with disease control in biologic treated type-2 dominant CRS. Int Forum Allergy Rhinol. In press.
-
Fokkens WJ, Lund VJ, Hopkins C, Hellings PW, Kern R, Reitsma S, et al. European Position Paper on Rhinosinusitis and Nasal Polyps 2020. Rhinology. 2020;58(Suppl S29):1-464.
-
Sedaghat AR, Fokkens WJ, Lund VJ, Hellings PW, Kern RC, Reitsma S, et al. Consensus criteria for chronic rhinosinusitis disease control: an international Delphi study. Rhinology. 2023;61(6):519-530.
-
Hopkins C, Gillett S, Slack R, Lund VJ, Browne JP. Psychometric validity of the 22-item Sinonasal Outcome Test. Clin Otolaryngol. 2009;34(5):447-454.
-
Bachert C, Han JK, Wagenmann M, Hosemann W, Lee SE, Backer V, et al. EUFOREA expert board meeting on uncontrolled severe chronic rhinosinusitis with nasal polyps (CRSwNP) and biologics: Definitions and management. J Allergy Clin Immunol. 2021;147(1):29-36.
-
Scangas GA, Wu AW, Ting JY, Metson R, Walgama E, Shrime MG, et al. Cost utility analysis of dupilumab versus endoscopic sinus surgery for chronic rhinosinusitis with nasal polyps. Laryngoscope. 2020.
-
Fieux M, Margier J, Bartier S, Chang M, Carsuzaa F, Hwang PH, et al. The extra cost of biologics as first-line treatment in uncontrolled chronic rhinosinusitis with nasal polyps with no previous sinus surgery is overwhelming: a budget impact analysis. Rhinology. 2025;63(4):495-504.